PharmaSources/YuntianMay 25, 2022
Tag: Cepharanthine , COVID-19 , Synthesis Technology
Recently, cepharanthine, first discovered in China as the new drug for treating COVID-19, has recently been granted the national invention patent authorization, which has drawn great attention from inside and outside the industry. In accordance with the authorized invention patent, "10uM cepharanthine can inhibit the replication of coronavirus by 15,393 times", and it's firmly remarkable and exciting for inhibition effect on coronavirus. Let's take a look at the following synthesis technology of this natural product.
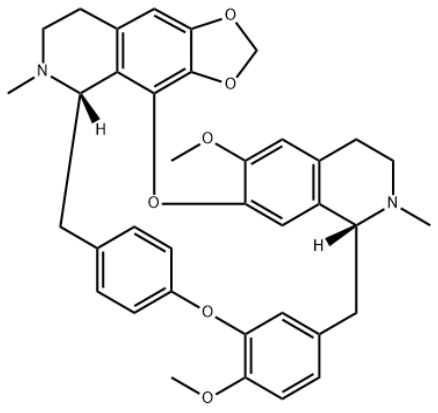
Figure 1 Structural formula of Cepharanthine
There is only one report on the synthesis technology of cepharanthine via retrieval, which is applied to synthesize the racemate of cepharanthine. As shown in Figure 2, the first one is to synthesize the intermediate 5. Aldehyde 1 is utilized as the starting material in this process, which reacts with dibromomethane to obtain intermediate 2. With the catalysis of ammonium acetate, intermediate 2 reacts with nitromethane to form intermediate 3. The intermediate 3 is reduced under the reduction conditions of Clemmensen to obtain amine compound 4. The intermediate 5 can be obtained by aldehyde reaction of compound 4.
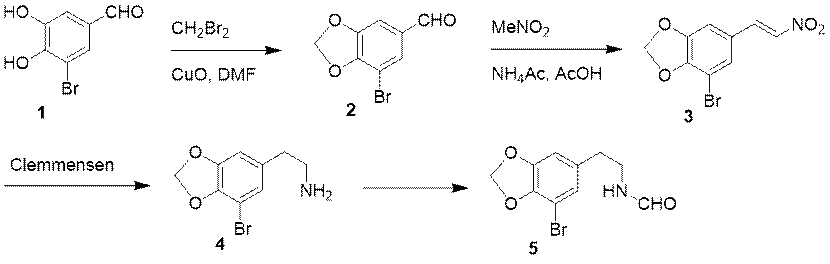
Figure 2 Synthesis of Intermediate 5
The researchers explored the synthesis of intermediate 9. Compound 6, as raw material, reacts with ethyl chloroformate to obtain intermediate 7. The intermediate 7 is reduced under the reduction conditions of Clemmensen to obtain amine compound 8. The amino group of compound 8 is under the protection of Cbz to obtain intermediate 9.
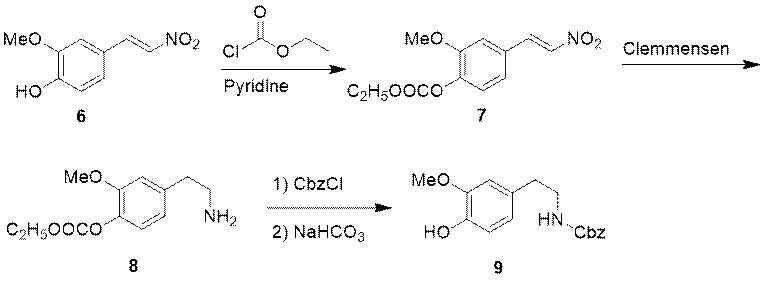
Figure 3 Synthesis of Intermediate 9
Next, the researchers made intermediate 5 to react with intermediate 9. With the methods of Ullmann condensation and the removal of the aldehyde group, the intermediate 10 can be obtained.
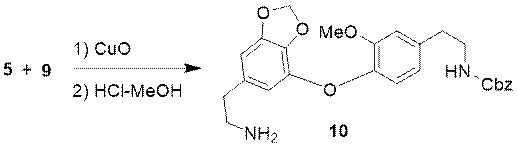
Figure 4 Synthesis of Intermediate 10
The intermediate 13, taking carboxylic acid 11 as the raw materials, can be synthesized by removing benzyl protection of intermediate 12, which can be obtained by esterification.
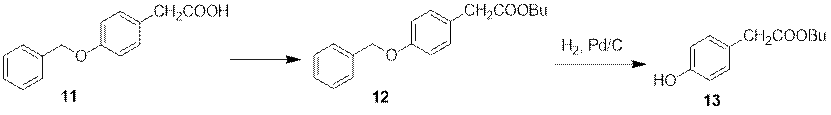
Figure 5 Synthesis of Intermediate 13
The intermediate 16 is synthesized with compound 14 as the raw materials, which firstly undergoes the methylation reaction with dimethyl sulfate under the catalysis of the potassium carbonate to obtain intermediate 15. After the Ullmann condensation reaction of intermediate 13 and 15, the intermediate 16 is obtained under the hydrolyzation of p-Toluenesulfonic acid.
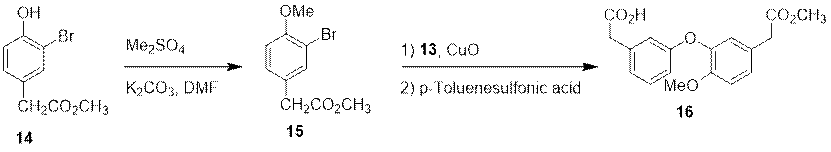
Figure 6 Synthesis of Intermediate 16
Based on the above intermediates, the researchers started the final synthesis of cepharanthine racemate. The intermediate 10 undergoes amide condensation reaction with intermediate 16 to obtain intermediate 17. The intermediate 17 is hydrolyzed in sodium carbonate to obtain the intermediate 17. The intermediate 17 undergoes cyclization reaction with the guidance of literature to obtain the intermediate 18. The intermediate 18 first undergoes Bischler-Napieralski reaction and then, is reduced to obtain cepharanthine racemate 19.
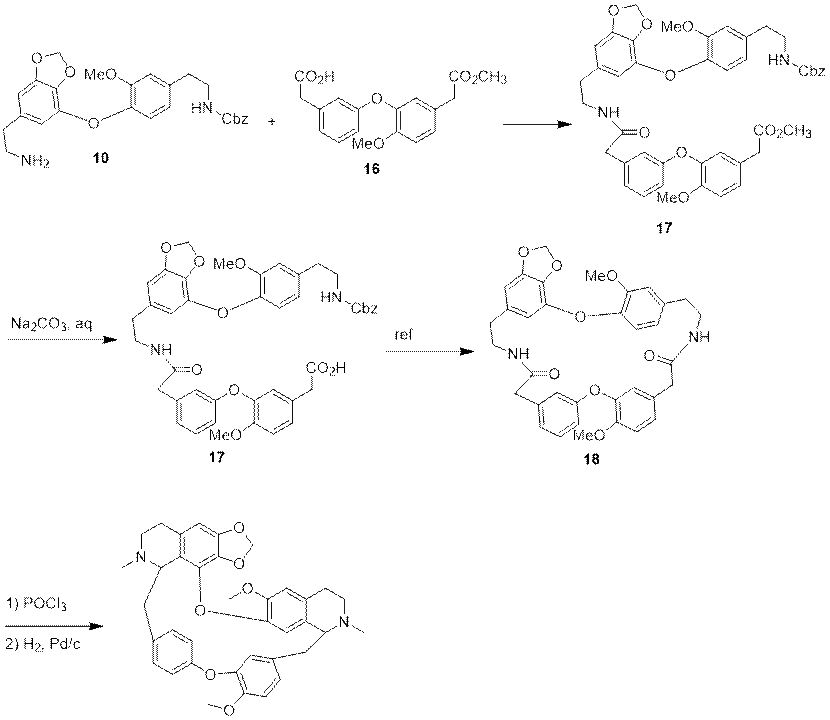
Figure 7 Synthesis of Cepharanthine Racemate
At present, cepharanthine is clinically applied as a leukocyte proliferation drug to treat leukopenia. It can promote the proliferation of bone marrow tissue, and then increase the number of leukocytes. Studies have proved that cepharanthine can increase the number of leukocytes in peripheral blood, markedly prevent leukopenia caused by mitomycin, and does not inhibit the antitumous effect of mitomycin.
In this time, it has proved that cepharanthine is featured with remarkable inhibitory effect on coronavirus, which is an unexpected effect. There is a great potential of new use for an old drug. However, the researchers said that it is far from being marketed as a coronavirus drug.
Although the total synthesis can provide an effective guarantee for the subsequent application of cepharanthine and the above process can be applied to obtain cepharanthine racemate, it is still far from being approved for industrial application because of such main limitations as long route, low total yield, and the synthesis of racemate instead of chiral products. Therefore, further development shall be conducted and more attention shall be paid to the application of the coronavirus infection treatment and the synthesis technology of cepharanthine.
1. Pangolin coronavirus xCoV and its application and application of drugs against coronavirus infection, CN 202010096868, invention patent;
2. Cepharanthine: a review of the antiviral potential of a Japanese-approved alopecia drug in COVID-19,2020。
Yuntian, Ph.D. in medicinal chemistry, is mainly engaged in small molecule drug research, especially good at small molecule drug synthesis process and later stage drug development research. He has completed the synthesis and activity evaluation of multiple anti-cancer drug molecules.


Contact Us
Tel: (+86) 400 610 1188
WhatsApp/Telegram/Wechat: +86 13621645194
+86 15021993094
Follow Us:




 Pharma Sources Insight July 2025
Pharma Sources Insight July 2025


